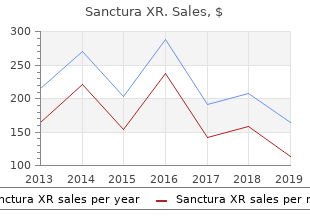
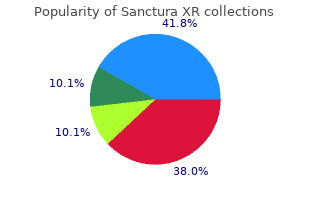
Sanctura XR
", symptoms for pregnancy."
By: Stephen M. Rosenthal MD
- Professor of Pediatrics, Associate Program Director, Pediatric Endocrinology
- Director, Pediatric Endocrine outpatient Services, University of California, San Francisco

https://profiles.ucsf.edu/stephen.rosenthal
In his opinion , 297 other possible etiological components embody hypertension and the patient�s age. Examples of Acceptable Company Clinical Evaluation Comments for Possible use in Paragraph eight of a Standard Narrative 1. The temporal relationship (6 weeks) between the onset of the event and administration of drug x, which has a one-hour half-life, makes any causal relationship unlikely. It is of curiosity to observe that the patient was subsequently rechallenged on the similar dose without recurrence of the opposed impact. The event resolved while drug x was continued on the similar dose which makes any relationship to the drug unlikely. The co-drugs y and z should also be thought of causative; the reported event is labeled for both medicine. The medicine was not administered in accordance with the dosage advice for the drug. The investigator on comply with-up has modified his evaluation from ��in all probability�� to ��in all probability not�� for the following causes �. This case has also been forwarded to (name of the opposite producer) as � (drug name) is the first suspect drug. The patient�s medical historical past offers an alternate explanation for the reported event. Examples of Unacceptable Company Clinical Evaluation Comments for Paragraph eight of a Standard Narrative 1. The investigator modified his evaluation from ��in all probability�� to ��in all probability not�� on comply with-up. Furthermore, for merchandise with subsequent additional regulatory approvals (new indications, new dosage varieties, etc. This has significant implications with regard to database minimize-off dates (knowledge lock-points), evaluation and presentation of information, in addition to for preparation and submission of reports (which is required not more than 60 days past the data lock-point date. However, this schedule will imply that for older merchandise not accredited through the centralized or mutual recognition procedures, reports on a single drug masking completely different time durations (6 months, one 12 months or 5 years of information) could also be required, possibly at completely different instances, in several international locations, relying on the approval dates in these international locations. For instance, the Finnish and Belgian agencies demand that the minimize-off date (knowledge lock-point) for a five 12 months report be within 6 months previous to the renewal date. Theoretically, if a global birthdate acceptable in all international locations could be established for all formulations of a drug, the five-12 months report could be compiled only once each 5 years when the product had reached maturity in all relevant international locations; regulators must conform to permit flexibility in earlier submissions relative to the local birthdates to allow synchroniza tion of reports for all regulators. At present, corporations are dealing with this situation in a number of alternative ways. Some corporations supplement their already ready five-12 months updates with line listings of reports masking the time between the minimize-off for the five-12 months report and the later submission. Others produce a sequence of 5-12 months reports that cover overlapping 5 12 months durations. The scenario turns into even more difficult if the reporting clock is set again to six-month-to-month when a new formulation or new indication is accredited for a drug already on or close to a 5 12 months reporting schedule.
In that sense , one should distinguish between helpful, sensible product data and regulatory reporting concerns. A minority of the Working Group instructed that, despite the fact that all expected (labelled, listed) drug-associated dying cases are routinely lined in periodic security reports, there could also be a public health argument for expedited reporting of all drug associated deaths. Thus, constant alert by way of expedited reporting of deaths, even if already labeled, was believed essential for public health and would offer confidence to the public that systems are in place for controlling such essential drug issues of safety. It is also instructed that the distinction between a life threatening medical scenario resulting in survival of the patient and an end result of dying might rely merely on the supply of acceptable medical care and experience; in that sense, any distinction made for expedited vs periodic reporting between ��life threatening�� and ��dying�� is synthetic. The majority felt that within the absence of particular circumstances, a more deliberate, 30 periodic evaluation is important for proper perspective. For instance, an oncology drug might not cure a hospitalised patient�s cancer, which results in a prolongation of hospitalisation. However, what if the treatment exacerbates the ��goal�� disease (the indication for the medicinal product) An instance of such a 121 paradoxical statement is bronchial asthma exacerbation caused, albeit rarely, by some bronchial asthma therapies. In this case, the bronchial asthma exacerbation would be surprising except detailed within the prescribing data. Similarly, if an anti-migraine treatment were adopted by an increase in frequency or depth of migraine attacks above baseline, then except listed within the security data, this adverse consequence would be surprising. Another vital concern is detection of an ��uncommon�� lack of expected therapeutic effect for medicines utilized in life-threatening ailments, which may have life or dying penalties. Does the looks of an ��uncommon�� incidence of reports from a number of sources indicate a signal of significance For this reason the criterion of ��elevated frequency�� of spontaneous cases is generally now not a prescribed, routine requirement for expedited reporting to regulatory agencies. One or more clusters of cases in localised areas or throughout a short time period will result in a search for a proof. A particular scenario arises when a sequence of individual cases might not have initially been thought of drug-associated, but upon separate evaluation. That constitutes a signal as properly and will require immediate notification to regulators. In common, statements involving frequency in product data should be thought of fastidiously and developed with full consideration of the difficulties in establishing denominators (publicity). A choice in many situations will have to be primarily based on clinical evaluation of insufficient case data.
. TLC Blood Orange Essential Oil!.

The function of early risk factors , family ecology, and treatment historical past on treatment response stays unclear. Treatment decision dilemmas are additional complicated for sufferers with medical and/or psychiatric comorbidities. This evaluation particularly centered on psychosocial and pharmacologic interventions for disruptive behavior. We included studies of father or mother-focused psychosocial interventions if the research reported adjustments to baby disruptive behavior. For pharmacologic interventions, we focused the literature on their use in disruptive behavior disorders, focusing on a smaller but extra centered literature base. The alternative of outcomes on which to focus the analysis and particularly the strength of proof was difficult for this evaluation. Many different measures are used to assess elements of disruptive behavior, not all of which have been validated. Excludes faculty extensive or system extensive settings wherein interventions are focused extra widely. Nonpharmacologic interventions normally are really helpful because the initial strategy, but clinicians and families are likely to use both approaches at some point, probably simultaneously, creating additional decisional dilemmas related to co remedy, polypharmacy, and the function of treatment historical past. We subsequently framed the Key Questions to verify the comparative effectiveness of various psychological and pharmacologic remedies aimed toward disruptive behaviors, in contrast both within and between treatment types, and verify whether there are mixtures of psychological and pharmacologic therapeutic approaches that are optimum. Key Question 1: In youngsters underneath 18 years of age handled for disruptive behaviors, are any psychosocial interventions more effective for enhancing quick-time period and lengthy-time period psychosocial outcomes than no treatment or other psychosocial interventions Key Question 5: What are the harms related to treating youngsters underneath 18 years of age for disruptive behaviors with either psychosocial or pharmacologic interventions Key Question 6a: Do interventions supposed to tackle disruptive behaviors and recognized in Key Questions 1-4 range in effectiveness primarily based on affected person traits, including sex, age, racial/ethnic minority, family historical past of disruptive behavior disorders, family historical past of psychological well being disorders, historical past of trauma, and socioeconomic standing Key Question 6b: Do interventions supposed to tackle disruptive behaviors and recognized in Key Questions 1-4 range in effectiveness primarily based on traits of the disorder, including specific disruptive behavior or disruptive behavior disorder. Key Question 6c: Do interventions supposed to tackle disruptive behaviors and recognized in Key Questions 1-4 range in effectiveness primarily based on treatment historical past of the affected person Analytic Framework the analytic framework (Figure 1) illustrates the population, interventions, outcomes, and adverse effects that may guide the literature search and synthesis. Analytic framework Organization of this Report We have organized the report by Key Question. For Key Question 1 (psychosocial interventions) we current the studies by age (categorized as preschool, faculty-age, and adolescent) and then additional divide the sections by single or multiple intervention elements.
The purpose was to mix good anti-shudder properties with the ability to lubricate hypoid gears, which meant that the addition of extreme strain components was essential. Base Oil Choice of base oil has only a minor effect on anti-shudder properties as could be seen in Figure 18, Figure 19 and Figure 20. This will assist improve fluid life since shearing of viscosity index improvers is understood to be a limiting factor on the fluid life in clutch applications. Single Additives After the base oil had been chosen, single additive checks were carried out in a reciprocating friction and wear tester, see Paper F. Initially, just one additive at the time was added to the base oil and the friction traits measured. With only a single additive within the base oil it was not possible to use the Limited Slip Clutch Test Rig as a result of stick-slip issues. While a few of the components studied similar to antioxidants, dispersants and detergents were found to have only minor influence on friction different, extra surface energetic components, showed significant influence as could be seen in Figure 24, Figure 25 and Figure 26. The performance of the components tested were ranked based mostly on the ratio between the static friction (peak value) and the dynamic friction. In addition to this, a excessive dynamic friction was sought in order to improve the torque transmission functionality of the clutch. The most promising components were selected for additional analysis in combination with different components within the subsequent stage of development. The friction modifier improved the anti-shudder performance significantly and the most effective friction modifier from the only additive testing was selected as the idea for the brand new formulation. Additive Combinations the second stage of development evaluated interactions between components. An initial formulation was outlined, consisting of an artificial base oil with antioxidant, friction modifier and detergent components. The analysis of additive combinations were divided into three steps and at every the first step type of additive launched. Selection of a combination to be developed additional was made by evaluating the friction-velocity curves and temperature dependence of friction. At step two and three, anti wear and excessive strain components respectively were added. In Figure 27 the compensated friction traits for the brand new fluid and the industrial fluid are plotted for a temperature of 50C. The friction traits (anti-shudder properties) are also good for both fluids even though the formulated fluid exhibits a slight unfavorable slope at greater velocities. Figure 28 and Figure 29 present friction as a perform of temperature for 1rpm (torque capacity) and 50rpm (dynamic friction) respectively. Both fluids have low temperature dependency at excessive sliding speeds whilst friction at low velocities is extra significantly influenced by temperature. This is to be anticipated for the reason that components, as shown earlier, have a powerful influence on friction especially at low velocities and the consequences of components. The new fluid had a larger temperature dependence than the industrial fluid especially at low velocities and at low temperatures.
